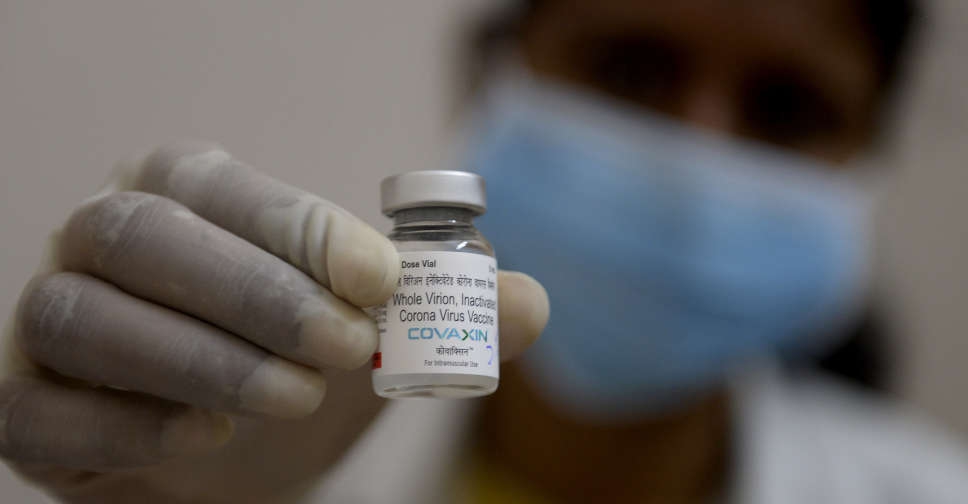
A World Health Organisation (WHO) technical advisory group was reviewing data on India's Covaxin shot against COVID-19 on Tuesday with a decision on its emergency use listing likely soon, a spokesperson said.
"If all is in place and all goes well and if the committee is satisfied, we would expect a recommendation within the next 24 hours or so," Margaret Harris told journalists at a UN press briefing.
Millions of Indians have taken the shot produced by Bharat Biotech but many have been unable to travel pending the WHO approval.




 WHO chief says 12 dead in strike on Lebanon's primary healthcare center
WHO chief says 12 dead in strike on Lebanon's primary healthcare center
 India seeks passage of more vessels after some pass through Strait of Hormuz
India seeks passage of more vessels after some pass through Strait of Hormuz
 US embassy in Baghdad hit in missile attack, security sources say
US embassy in Baghdad hit in missile attack, security sources say
 US orders non-emergency personnel to leave Oman amid regional conflict
US orders non-emergency personnel to leave Oman amid regional conflict
 US identifies six service members killed in plane crash over Iraq
US identifies six service members killed in plane crash over Iraq
 Israel threatens to strike ambulances in Lebanon in Hezbollah fighting
Israel threatens to strike ambulances in Lebanon in Hezbollah fighting
 Death toll from Kenyan floods rises to 62, police say
Death toll from Kenyan floods rises to 62, police say
 Trump threatens to strike Iran's Kharg Island oil network
Trump threatens to strike Iran's Kharg Island oil network




