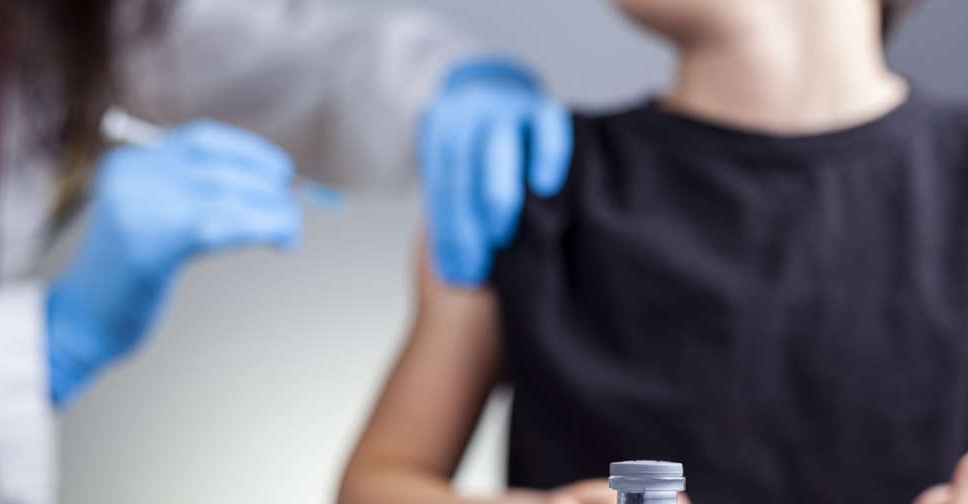
The UAE's Ministry of Health and Prevention has given emergency approval for Sinopharm COVID-19 vaccine for children aged three and above.
In a tweet, the National Emergency Crisis and Disasters Management Authority (NCEMA) said the decision was made after "the recent clinical trials and extensive evaluations, which are in line with the approved regulations".
Ministry of Health approves use of #Sinopharm vaccine for 3-17 age group #WamNews pic.twitter.com/yq8snmccJu
— WAM English (@WAMNEWS_ENG) August 2, 2021
MoHAP announced the provision of Sinopharm vaccine for the age group 3 - 17 The decision comes after clinical trials and extensive evaluations and is based on the emergency use authorization and local evaluations which are in line with the approved regulations.#TogetherWeRecover pic.twitter.com/4Y3VEMMW8V
— NCEMA UAE (@NCEMAUAE) August 2, 2021
It comes after authorities in Abu Dhabi achieved the target of vaccinating 900 children as part of a trial to study the effectiveness of the Sinopharm COVID-19 vaccine in young people.
The Department of Health Abu Dhabi (DoH) said the study examined the vaccine’s effectiveness in reducing the infection rate and severity of symptoms among target groups.
Its preliminary results will be announced at a later stage.




 UAE leaders highlight unity and strength in Dubai meeting
UAE leaders highlight unity and strength in Dubai meeting
 UAE modifies alert sounds in National Early Warning System
UAE modifies alert sounds in National Early Warning System
 2 members of UAE Armed Forces martyred in helicopter crash
2 members of UAE Armed Forces martyred in helicopter crash
 UAE condemns drone attack on consulate in Iraqi Kurdistan
UAE condemns drone attack on consulate in Iraqi Kurdistan
 UAE participates in extraordinary EU–GCC leaders meeting
UAE participates in extraordinary EU–GCC leaders meeting
 Spanish PM reaffirms support for UAE following Iranian attacks
Spanish PM reaffirms support for UAE following Iranian attacks
 UAE welcomes US listing of Sudanese Muslim Brotherhood as terrorist group
UAE welcomes US listing of Sudanese Muslim Brotherhood as terrorist group
 UAE ambassador to UN calls for 'de-escalation' in Iran conflict
UAE ambassador to UN calls for 'de-escalation' in Iran conflict




