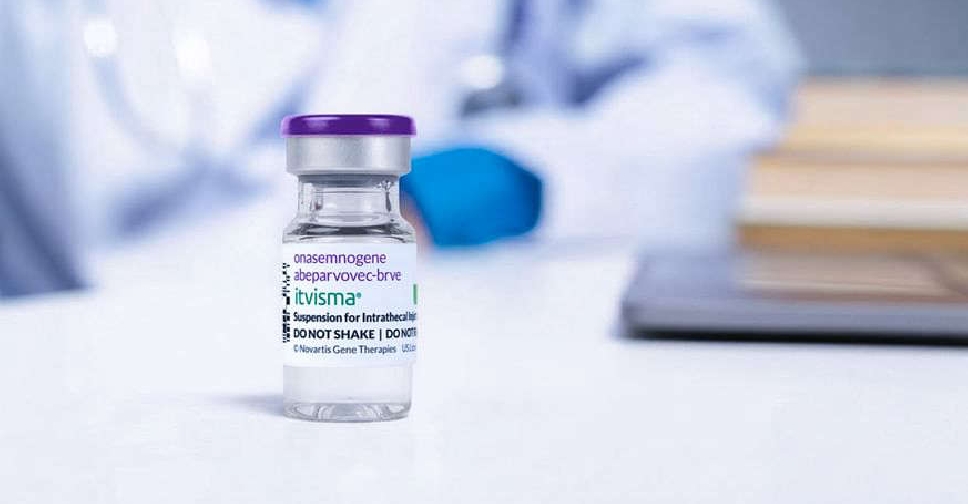
Abu Dhabi has become the first in the world to deliver Itvisma, a one-time gene therapy specifically designed to target the underlying genetic cause of spinal muscular atrophy (SMA) in patients aged two years and above.
Sheikh Khalifa Medical City (SKMC) has successfully administered this groundbreaking gene therapy, developed by Novartis, under the supervision of the Department of Health – Abu Dhabi (DoH), the regulator of the healthcare sector in the emirate.
The therapy had been previously approved by the UAE on November 25, positioning it among the first countries globally, after the USA, to endorse the pioneering treatment.
Designed for simplicity long-term impact, Itvisma (onasemnogene abeparvovec) replaces the missing SMN1 gene to improve motor function, reducing the need for continuous treatments required by other therapies.
Dr. Noura Khamis Al Ghaithi, Under-Secretary of DoH, stated that “this milestone reflects Abu Dhabi’s commitment to delivering world class care and strengthening its position as a global leader in healthcare driven by genomics and precision medicine."



 UAE participates in GCC Consultative Summit in Saudi Arabia
UAE participates in GCC Consultative Summit in Saudi Arabia
 UAE President to honour Abdullah Al Sayed Al Hashemi with Abu Dhabi street name
UAE President to honour Abdullah Al Sayed Al Hashemi with Abu Dhabi street name
 Sharjah Police arrest suspect accused of turning home into drugs 'factory'
Sharjah Police arrest suspect accused of turning home into drugs 'factory'
 H.H. Sheikh Mohammed renames Ministerial Development Council
H.H. Sheikh Mohammed renames Ministerial Development Council
 Dubai establishes framework to boost consumer rights in aviation sector
Dubai establishes framework to boost consumer rights in aviation sector
 Abu Dhabi hosts daily flag ceremony to honour martyrs
Abu Dhabi hosts daily flag ceremony to honour martyrs
 Sharjah hands out AED 9.14 million in annual bonuses to old taxi plate owners
Sharjah hands out AED 9.14 million in annual bonuses to old taxi plate owners
 H.H. Sheikh Mohammed visits world's largest private dates factory at DIC
H.H. Sheikh Mohammed visits world's largest private dates factory at DIC




