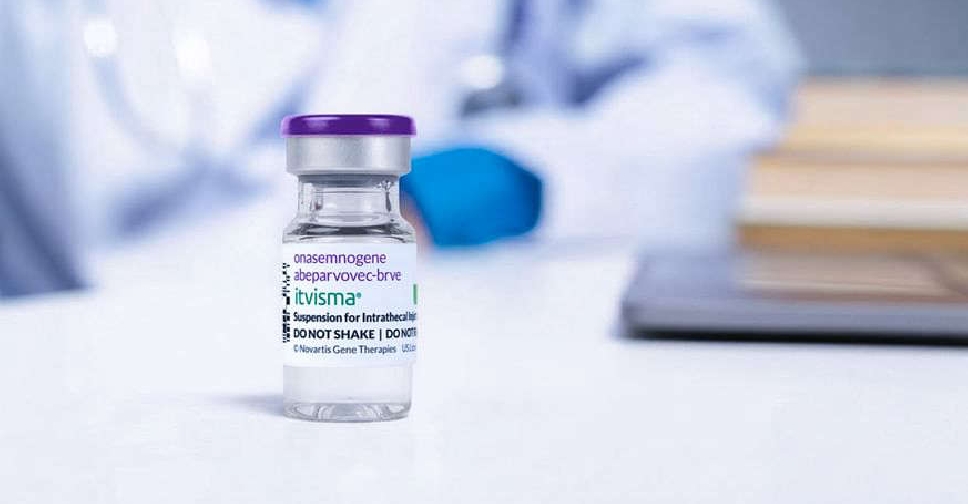
Abu Dhabi has become the first in the world to deliver Itvisma, a one-time gene therapy specifically designed to target the underlying genetic cause of spinal muscular atrophy (SMA) in patients aged two years and above.
Sheikh Khalifa Medical City (SKMC) has successfully administered this groundbreaking gene therapy, developed by Novartis, under the supervision of the Department of Health – Abu Dhabi (DoH), the regulator of the healthcare sector in the emirate.
The therapy had been previously approved by the UAE on November 25, positioning it among the first countries globally, after the USA, to endorse the pioneering treatment.
Designed for simplicity long-term impact, Itvisma (onasemnogene abeparvovec) replaces the missing SMN1 gene to improve motor function, reducing the need for continuous treatments required by other therapies.
Dr. Noura Khamis Al Ghaithi, Under-Secretary of DoH, stated that “this milestone reflects Abu Dhabi’s commitment to delivering world class care and strengthening its position as a global leader in healthcare driven by genomics and precision medicine."



 UAE responds to further missile, drone threats on Sunday
UAE responds to further missile, drone threats on Sunday
 16 ballistic missiles, 42 drones intercepted on Sunday
16 ballistic missiles, 42 drones intercepted on Sunday
 Gargash: Political solution needs guarantees against repeat Iranian attacks
Gargash: Political solution needs guarantees against repeat Iranian attacks
 H.H. Sheikh Mohammed congratulates Dubai World Cup winner
H.H. Sheikh Mohammed congratulates Dubai World Cup winner
 Sheikh Abdullah discusses impact of Iranian attacks with global partners
Sheikh Abdullah discusses impact of Iranian attacks with global partners
 Dubai opens 13 new cycling tracks under 162km RTA expansion plan
Dubai opens 13 new cycling tracks under 162km RTA expansion plan
 UAE condemns targeting of Kurdistan President's residence
UAE condemns targeting of Kurdistan President's residence
 Emirates Global Aluminium reports significant damage after attack
Emirates Global Aluminium reports significant damage after attack




