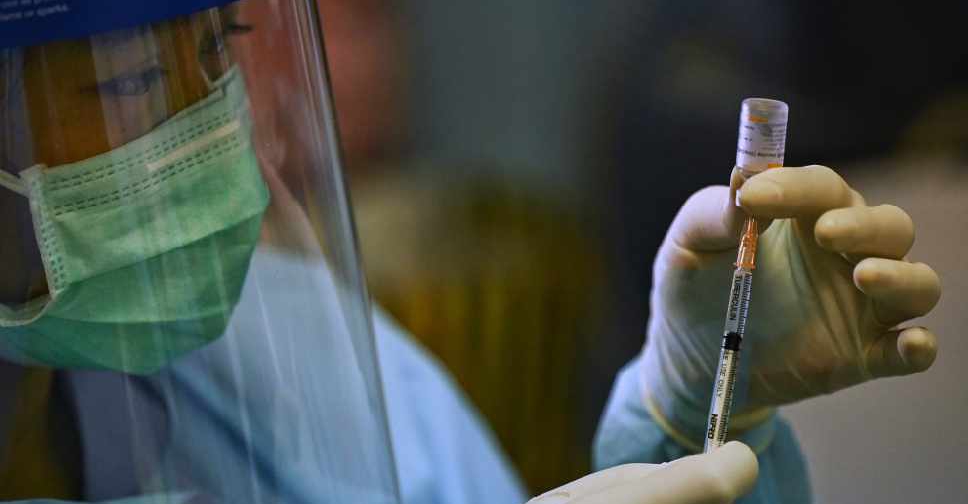
The World Health Organisation (WHO) has approved a COVID-19 vaccine made by Sinovac Biotech for emergency use listing.
It's the second Chinese-produced shot to get its endorsement, a WHO statement said on Tuesday.
WHO emergency listing is a signal to national regulators on a product's safety and efficacy.
It would also allow the shot to be included in COVAX, the global programme to provide vaccines mainly for poor countries, which currently faces major supply problems.
In a statement, the independent panel of experts said it recommended Sinovac's vaccine for adults over 18, with a second dose 2-4 weeks later.
There was no upper age limit as data suggested it is likely to have a protective effect in older people.




 Iranian missile strikes oil tanker in Qatar
Iranian missile strikes oil tanker in Qatar
 Australia's Albanese says war's economic shock will be felt for months
Australia's Albanese says war's economic shock will be felt for months
 Iranian drones target fuel tanks at Kuwait International Airport
Iranian drones target fuel tanks at Kuwait International Airport
 At least 18 dead after migrant boat sinks off Turkey's Aegean coast
At least 18 dead after migrant boat sinks off Turkey's Aegean coast
 Trump says US to leave Iran 'very soon', will address nation Wednesday
Trump says US to leave Iran 'very soon', will address nation Wednesday
 Rubio says US can see 'finish line' on Iran war
Rubio says US can see 'finish line' on Iran war
 Fire breaks out at company facility in Bahrain after Iranian attack
Fire breaks out at company facility in Bahrain after Iranian attack
 Tehran threatens US businesses, Hegseth says next few days 'decisive'
Tehran threatens US businesses, Hegseth says next few days 'decisive'




