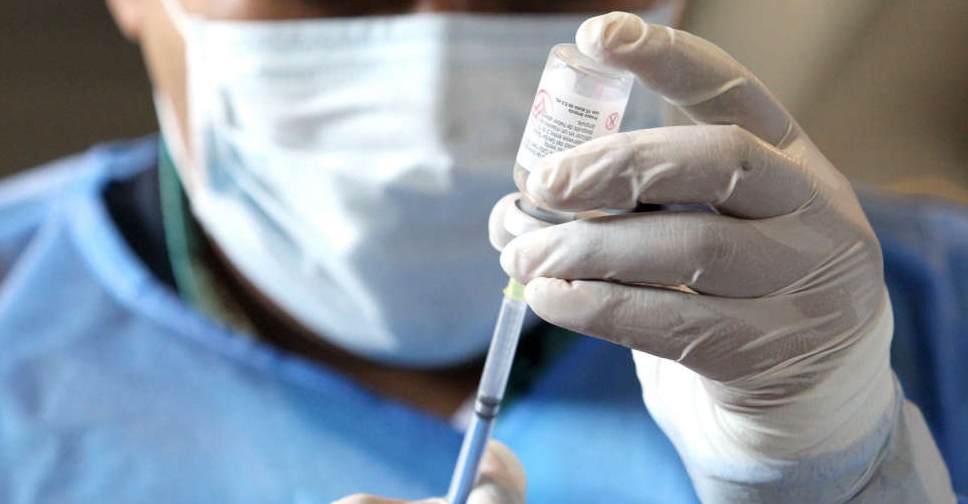
The World Health Organization (WHO) on Thursday issued an emergency use listing for the single-dose COVID-19 vaccine from China-based CanSino Biologics.
The vaccine, Convidecia, is the eleventh shot against the coronavirus to get clearance from the global health agency, whose advisory group recommended its use in people of age 18 years and above.
It was found to have 64 per cent efficacy against symptomatic disease and 92 per cent against severe COVID-19, the agency said.
Other vaccines that have similar clearance from the agency include those made by Pfizer and BioNTech, Moderna, AstraZeneca, Johnson & Johnson and Novavax.




 GCC interior ministers hold emergency meeting in Riyadh
GCC interior ministers hold emergency meeting in Riyadh
 Gunshots fired in standoff at Philippine Senate over ICC suspect
Gunshots fired in standoff at Philippine Senate over ICC suspect
 Trump lands in China for Xi summit
Trump lands in China for Xi summit
 Israeli airstrikes kill eight people on highway south of Beirut
Israeli airstrikes kill eight people on highway south of Beirut
 Philippines' Senator says arrest imminent, urges public to block ICC transfer
Philippines' Senator says arrest imminent, urges public to block ICC transfer
 UK's Starmer faces biggest challenge yet as resignation threat overshadows King's Speech
UK's Starmer faces biggest challenge yet as resignation threat overshadows King's Speech
 Israel steps up attacks on Gaza since Iran truce, as military says Hamas rearming
Israel steps up attacks on Gaza since Iran truce, as military says Hamas rearming
 Trump says he does not need China's help to end Iran war
Trump says he does not need China's help to end Iran war




