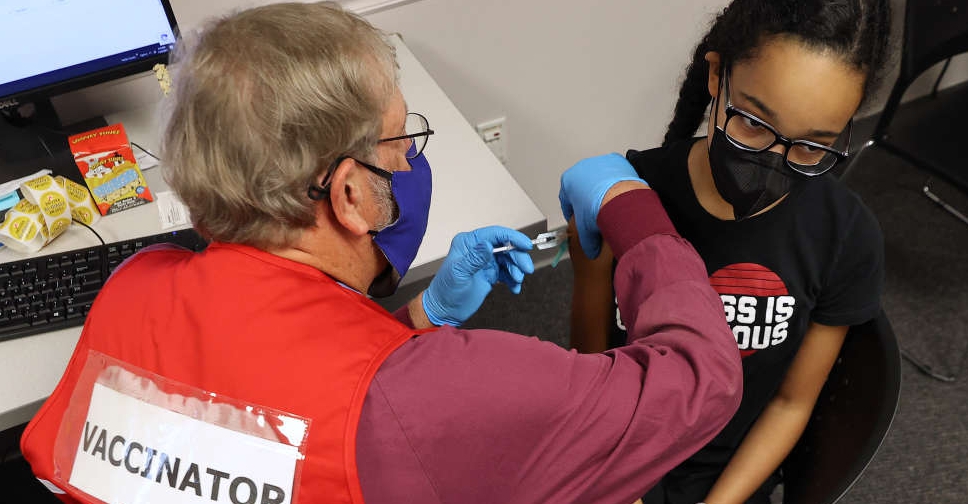
The US Food and Drug Administration (FDA) has authorised the use of a third dose of the Pfizer and BioNTech COVID-19 vaccine for children aged between 12 and 15 years.
The agency has also narrowed the time for all booster shots to 5 months from 6 months after primary doses.
Meanwhile, a third shot has been authorised in children aged 5 through 11 years who are immunocompromised.
The FDA said it reviewed published data and real world evidence on the safety of booster doses provided by the Israeli Ministry of Health including data from over 6,300 individuals 12-to-15 years of age who received a Pfizer shot.
Global COVID-19 cases are surging due to the Omicron variant and health authorities have warned that its extremely high transmissibility could overwhelm many health systems.
Laboratory tests have shown that two doses of the Pfizer-BioNTech and Moderna vaccines generate low immune responses against Omicron, while boosters appear to be protective against the highly-mutated variant.




 Israeli fire kills four in Gaza and the West Bank, medics say
Israeli fire kills four in Gaza and the West Bank, medics say
 Spain closes airspace to US planes involved in Iran war, El Pais says
Spain closes airspace to US planes involved in Iran war, El Pais says
 UN peacekeeper killed in southern Lebanon after projectile explosion
UN peacekeeper killed in southern Lebanon after projectile explosion
 Iranian attack kills one in Kuwait, damages distillation plant
Iranian attack kills one in Kuwait, damages distillation plant
 Iran accuses US of ground assault plans as Pakistan hosts regional talks
Iran accuses US of ground assault plans as Pakistan hosts regional talks
 Heavy rain, floods kill 22 people in Afghanistan
Heavy rain, floods kill 22 people in Afghanistan
 Fugitive accused of killing two Australian police officers shot dead
Fugitive accused of killing two Australian police officers shot dead
 Netanyahu orders expansion of security buffer zone in southern Lebanon
Netanyahu orders expansion of security buffer zone in southern Lebanon




