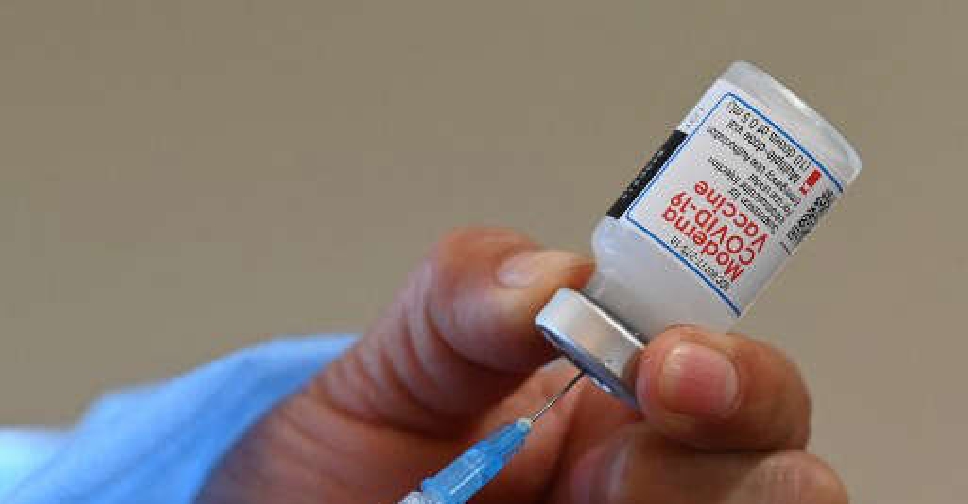
Britain's health regulator has approved Moderna's COVID-19 vaccine for use in children aged 12 to 17 years.
The approval comes more than two months after Pfizer and German partner BioNTech's COVID-19 vaccine got regulatory nod for use in children aged 12 to 15.
Moderna's vaccine was recommended for use in adolescents by European regulators in July and is awaiting US authorisation.
It is currently approved for people over the age of 18 in the UK.
Britain's Joint Committee on Vaccination and Immunisation (JCVI) gave the go-ahead on August 4 for 16 and 17-year-olds to get their first dose of Pfizer's COVID-19 vaccine ahead of schools returning in September.
JCVI will make a decision on whether the vaccine will be deployed or not.




 Trump dismisses Iran's reply to peace plan, oil jumps as Hormuz closure persists
Trump dismisses Iran's reply to peace plan, oil jumps as Hormuz closure persists
 Suspect in Trump attempted assassination pleads not guilty
Suspect in Trump attempted assassination pleads not guilty
 UK's Starmer promises to be bolder to try to rescue his job
UK's Starmer promises to be bolder to try to rescue his job
 Evacuation of passengers from virus-hit cruise ship to be completed on Monday
Evacuation of passengers from virus-hit cruise ship to be completed on Monday
 Philippine lawmakers to vote on impeachment of presidential hopeful Duterte
Philippine lawmakers to vote on impeachment of presidential hopeful Duterte
 Six people found dead in boxcar in Texas, police say
Six people found dead in boxcar in Texas, police say
 Turkish Airlines plane evacuated due to tyre fire after landing in Kathmandu
Turkish Airlines plane evacuated due to tyre fire after landing in Kathmandu
 Thailand's former PM Thaksin Shinawatra released from prison
Thailand's former PM Thaksin Shinawatra released from prison




