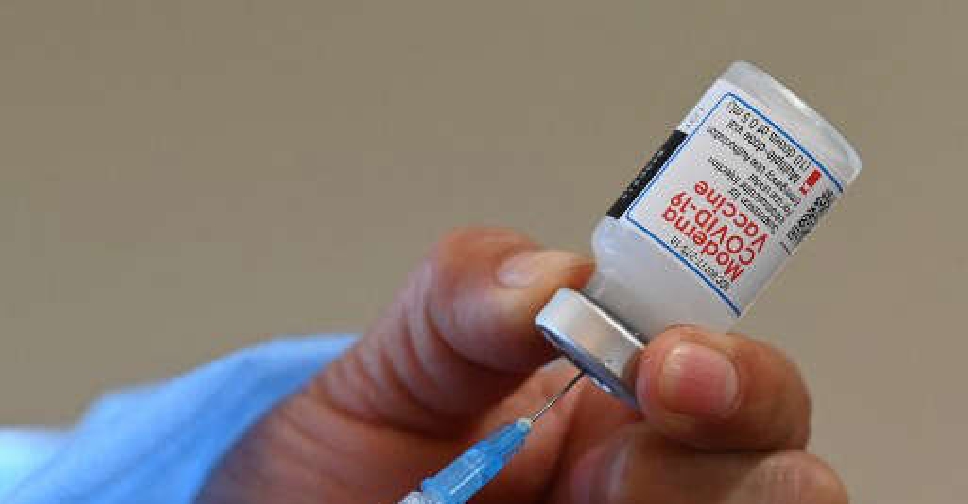
Britain's health regulator has approved Moderna's COVID-19 vaccine for use in children aged 12 to 17 years.
The approval comes more than two months after Pfizer and German partner BioNTech's COVID-19 vaccine got regulatory nod for use in children aged 12 to 15.
Moderna's vaccine was recommended for use in adolescents by European regulators in July and is awaiting US authorisation.
It is currently approved for people over the age of 18 in the UK.
Britain's Joint Committee on Vaccination and Immunisation (JCVI) gave the go-ahead on August 4 for 16 and 17-year-olds to get their first dose of Pfizer's COVID-19 vaccine ahead of schools returning in September.
JCVI will make a decision on whether the vaccine will be deployed or not.




 US proposes plan to end Iran war, as Trump signals progress
US proposes plan to end Iran war, as Trump signals progress
 US expected to send thousands more soldiers to Middle East
US expected to send thousands more soldiers to Middle East
 US safety agency says tracking system failed at LaGuardia during jet collision
US safety agency says tracking system failed at LaGuardia during jet collision
 Russia fires record 948 drones against Ukraine over 24 hours, Kyiv says
Russia fires record 948 drones against Ukraine over 24 hours, Kyiv says
 Death toll rises to 69 in Colombian military plane crash
Death toll rises to 69 in Colombian military plane crash
 One killed, five injured in Bahrain from Iran strikes
One killed, five injured in Bahrain from Iran strikes
 Qatar is not directly mediating between US and Iran, ministry spokesperson says
Qatar is not directly mediating between US and Iran, ministry spokesperson says
 Germany, France in rare rebuke of Trump over Iran war
Germany, France in rare rebuke of Trump over Iran war




