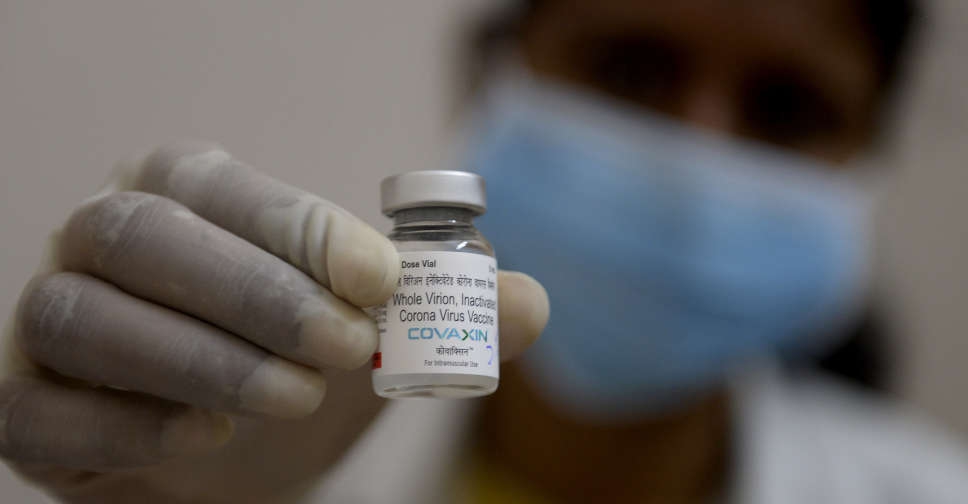
A World Health Organisation (WHO) technical advisory group was reviewing data on India's Covaxin shot against COVID-19 on Tuesday with a decision on its emergency use listing likely soon, a spokesperson said.
"If all is in place and all goes well and if the committee is satisfied, we would expect a recommendation within the next 24 hours or so," Margaret Harris told journalists at a UN press briefing.
Millions of Indians have taken the shot produced by Bharat Biotech but many have been unable to travel pending the WHO approval.




 UN establishes task force to address Hormuz navigation challenges
UN establishes task force to address Hormuz navigation challenges
 Iran-linked hackers breach FBI director's personal email
Iran-linked hackers breach FBI director's personal email
 Trump extends deadline for striking Iran's energy plants to April 7
Trump extends deadline for striking Iran's energy plants to April 7
 Israeli evacuation orders uproot 370,000 children in Lebanon, UN says
Israeli evacuation orders uproot 370,000 children in Lebanon, UN says
 UN rights chief urges US to conclude probe into deadly Iran school strike
UN rights chief urges US to conclude probe into deadly Iran school strike
 No injuries reported as Kuwait Shuwaikh Port comes under drone attack
No injuries reported as Kuwait Shuwaikh Port comes under drone attack
 IRGC-linked cells targeting Bahrain referred to Criminal Court
IRGC-linked cells targeting Bahrain referred to Criminal Court
 Austria plans social media ban for children under 14
Austria plans social media ban for children under 14




