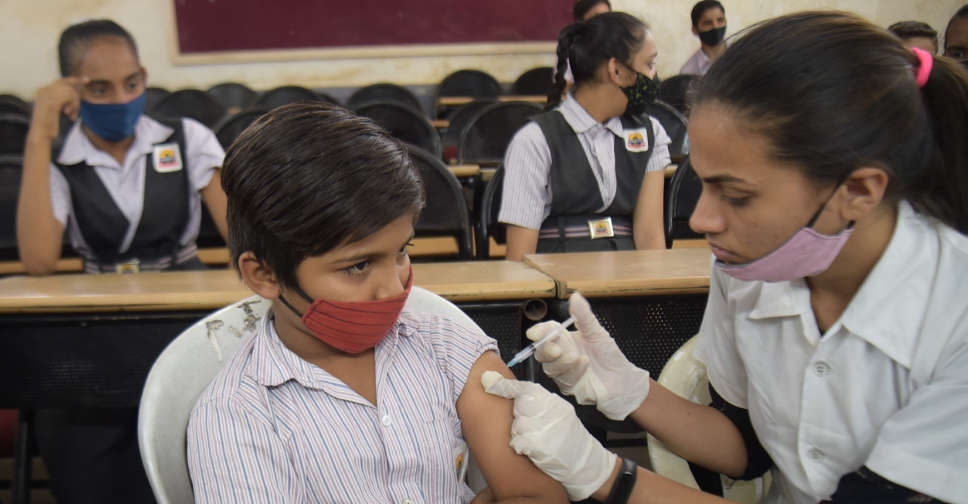
Novavax Inc said on Tuesday its COVID-19 vaccine has got emergency-use authorization from the Drugs Controller General of India for children aged 12 to 17 years.
The authorisation is a global first for the age group for the vaccine, which is manufactured and marketed in India by the Serum Institute of India under the brand name Covovax.
Novavax last month said its vaccine was 80 per cent effective against COVID-19 in a late-stage trial testing the shot in 2,247 teens aged 12 to 17 years. Read full story
The company said on Tuesday its vaccine produced an immune response in the same age group in a mid- to late-stage study involving 460 Indian adolescents.
Covovax is the fourth COVID-19 vaccine to be authorised for adolescents aged 12 years and older in India after Corbevax, ZyCoV-D and Covaxin.
India, which had so far been vaccinating children aged 15 and above, started administering doses of Corbevax last week to children aged 12 to 14.
The country's drug regulator in December authorised Novavax's COVID-19 vaccine for people aged 18 years and above.




 Iran denies talks with US after Trump postpones strikes on power grid
Iran denies talks with US after Trump postpones strikes on power grid
 Ukraine's Zelenskyy says Russia preparing new massive attack
Ukraine's Zelenskyy says Russia preparing new massive attack
 Rockets launched from Iraq’s Mosul towards US base in Syria
Rockets launched from Iraq’s Mosul towards US base in Syria
 One killed, 77 hospitalised after Colombian military plane crashes
One killed, 77 hospitalised after Colombian military plane crashes
 Hong Kong police given new powers to obtain phone, computer passwords
Hong Kong police given new powers to obtain phone, computer passwords
 ICE agents begin deploying at some US airports
ICE agents begin deploying at some US airports
 Air Canada jet collision shuts LaGuardia; pilots killed, dozens injured
Air Canada jet collision shuts LaGuardia; pilots killed, dozens injured
 Kuwait launches probe into espionage, state security cases
Kuwait launches probe into espionage, state security cases




