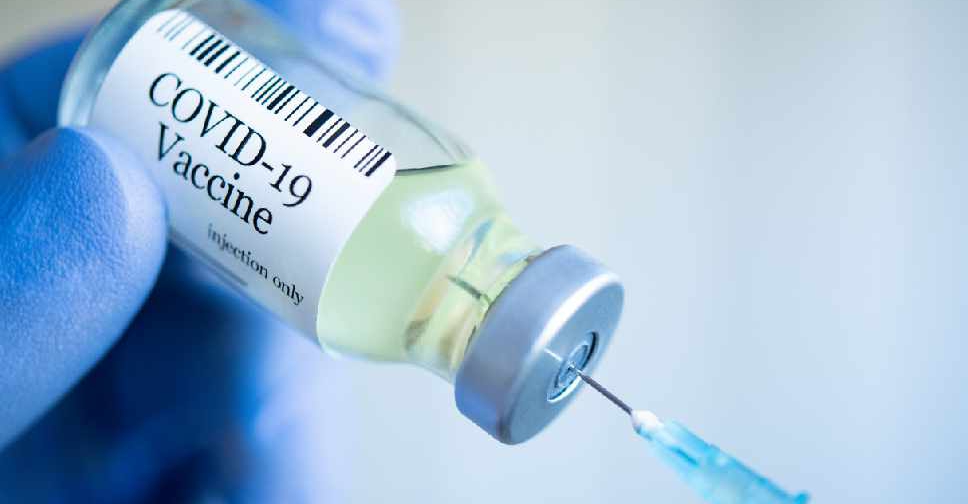
Sinovac Biotech said on Saturday that its unit's COVID-19 vaccine has been formally approved for use by the general public by China's medical products regulator.
It marks the second COVID-19 vaccine green-lighted for public use in China, after a shot developed by a Beijing institute affiliated to state-owned China National Pharmaceutical Group (Sinopharm) was approved in December.
Prior to the approvals, both vaccines have already been used in China's vaccination program mainly targeting key groups deemed to be at higher risk of exposure to the virus.
Indonesia, Turkey, Brazil, Chile, Colombia, Uruguay, and Laos have granted emergency authorizations for the CoronaVac vaccine developed by Sinovac Life Sciences, Sinovac said in a news release.
The approval is based on the two-month results from late-stage clinical trials overseas, from which the final analysis data has not yet been obtained, Sinovac said.




 US war in Iran has cost $29 billion so far, Pentagon says
US war in Iran has cost $29 billion so far, Pentagon says
 At Temple of Heaven summit, Trump and Xi will seek a good harvest
At Temple of Heaven summit, Trump and Xi will seek a good harvest
 Peace deal hopes fade after Trump rejects Iran proposal
Peace deal hopes fade after Trump rejects Iran proposal
 Protests erupt after key college entrance test cancelled in India
Protests erupt after key college entrance test cancelled in India
 UK's Starmer defies calls to quit, says he is getting on with governing
UK's Starmer defies calls to quit, says he is getting on with governing
 New Israeli law sets military tribunal for those linked to October 7 attack
New Israeli law sets military tribunal for those linked to October 7 attack
 Russia strikes Ukraine with drones as ceasefire ends
Russia strikes Ukraine with drones as ceasefire ends
 Dutch hospital quarantines 12 over breach of hantavirus protocol
Dutch hospital quarantines 12 over breach of hantavirus protocol




